Abcam ab214039
Transferrin receptor (TFRC, or TFR1, or CD71 in the FACS people textbooks) is one of the most important iron-related proteins. In our laboratory, we have used several anti TFRC antibodies for in vivo experiments. Good results in mouse spleens were obtained using either Abcam ab61134 (rabbit polyclonal) or the mouse monoclonal Thermo Fisher 13-6800. Both these antibodies react with the intracellular part of TFRC protein.
In 2020, we turned our attention to transferrin receptor protein circulating in plasma. Plasma transferrin receptors are derived from reticulocyte transferrin receptors, which are removed from the reticulocytes in exosomes – small vesicles of about 100 nm, which contain some reticulocyte membrane proteins. Although Professor Rose Johnstone discovered the removal of reticulocyte TFRC through exosomes in 1987, she is only rarely cited in iron-related literature. We speculated that, if TFRC is indeed shed in exosomes, then plasma exosomes from erythropoietin-treated mice should contain more TFRC than exosomes from control mice.
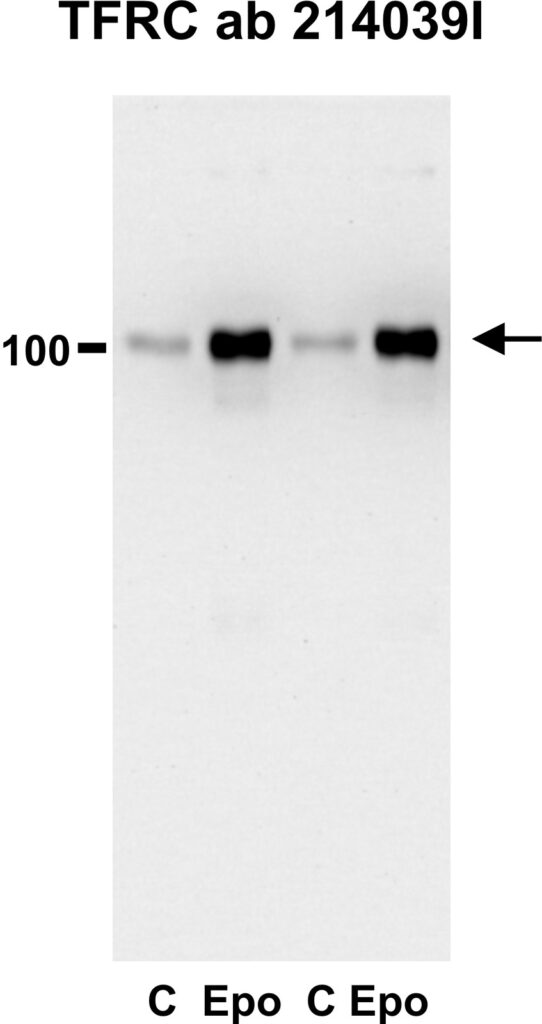
Our results obtained with the Abcam 214039 antibody show that exosomal TFRC dramatically increases in exosomes from erythropoietin-treated mice (exosomes were prepared by ultracentrifugation).
Seems Professor Johnstone was right!
The exosomal TFRC has a size around 100 kDa, indicating that it indeed is a membrane-bound, full-length protein. Note that in the erythropoietin-treated exosomes there is a smear at about 80 kDa: This probably represents contamination by cleaved soluble transferrin receptor from plasma.